Build, Measure, & Learn
Latest News from Pilloxa
iZafe signs a letter of intent to acquire Pilloxa AB
iZafe Group AB (publ) has signed a letter of intent with Pilloxa AB regarding the acquisition of 100 per cent of the shares in Pilloxa. Pilloxa has developed a technology platform for better compliance that pharmaceutical companies use to design digital patient support. The acquisition creates a new company in digital health that is well…
Continue Reading iZafe signs a letter of intent to acquire Pilloxa AB
Supporting Patients with Addison’s Disease and Type I Diabetes, an Interview with Dr. Dimitrios Chantzichristos
This article is the first in a series of pieces about Pilloxa and Sahlgrenska’s collaboration. Our Head of Marketing, Alexa Edstrom, sat down with research head Dr. Dimitrios Chantzichristos to discuss his upcoming research project and the use of digital components to support it. When a curious researcher approached Pilloxa in 2021 about our solutions,…
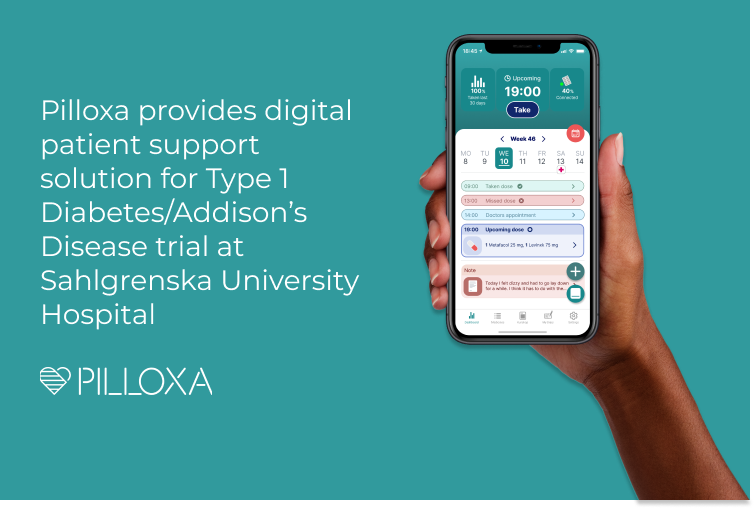
Pilloxa provides digital patient support solution for Type 1 Diabetes/Addison’s Disease trial at Sahlgrenska University Hospital
Stockholm, Sweden, April 26, 2022: Digital health company Pilloxa today announced they have signed a collaboration to provide a digital patient support solution based on their software and smart pillbox for a trial to be conducted at Sahlgrenska University Hospital involving patients with Type 1 Diabetes and Addison’s Disease. The project involves the Department of…
EUROSTARS supported ASTORIA study using Pilloxa’s digital medication adherence solution in cardiovascular disease patients begins
Stockholm, Sweden, March 9, 2022: Digital health company Pilloxa today announced that the First Patient First Visit milestone has been reached in the ASTORIA* study – a single-armed and multicenter observational clinical trial in which their digital patient-centric adherence solution is being tested in cardiovascular disease patients, namely newly diagnosed patients with atrial fibrillation. Professor…
Medication Adherence and the Consequences of Poor Adherence
Introduction and definition Adherence to medication is a term used to describe how well patients take their medication according to the plan created together with their healthcare professional. Sometimes it also includes adherence to other medical advice such as attending physical care, diet and lifestyle interventions, etc. The word adherence has come to replace the…
Continue Reading Medication Adherence and the Consequences of Poor Adherence
Pilloxa and the Nordic office of Chiesi Global Rare Diseases in collaboration to support patients with the rare disease nephropathic cystinosis across Scandinavia
Swedish digital startup company Pilloxa today announced that it is to collaborate with the Nordic office of Chiesi Global Rare Diseases on using the Pilloxa platform to support the treatment journey for patients with nephropathic cystinosis across Scandinavia. Nephropathic cystinosis is an ultra-rare, multisystemic, genetic disorder characterized by the accumulation of the amino acid cystine…
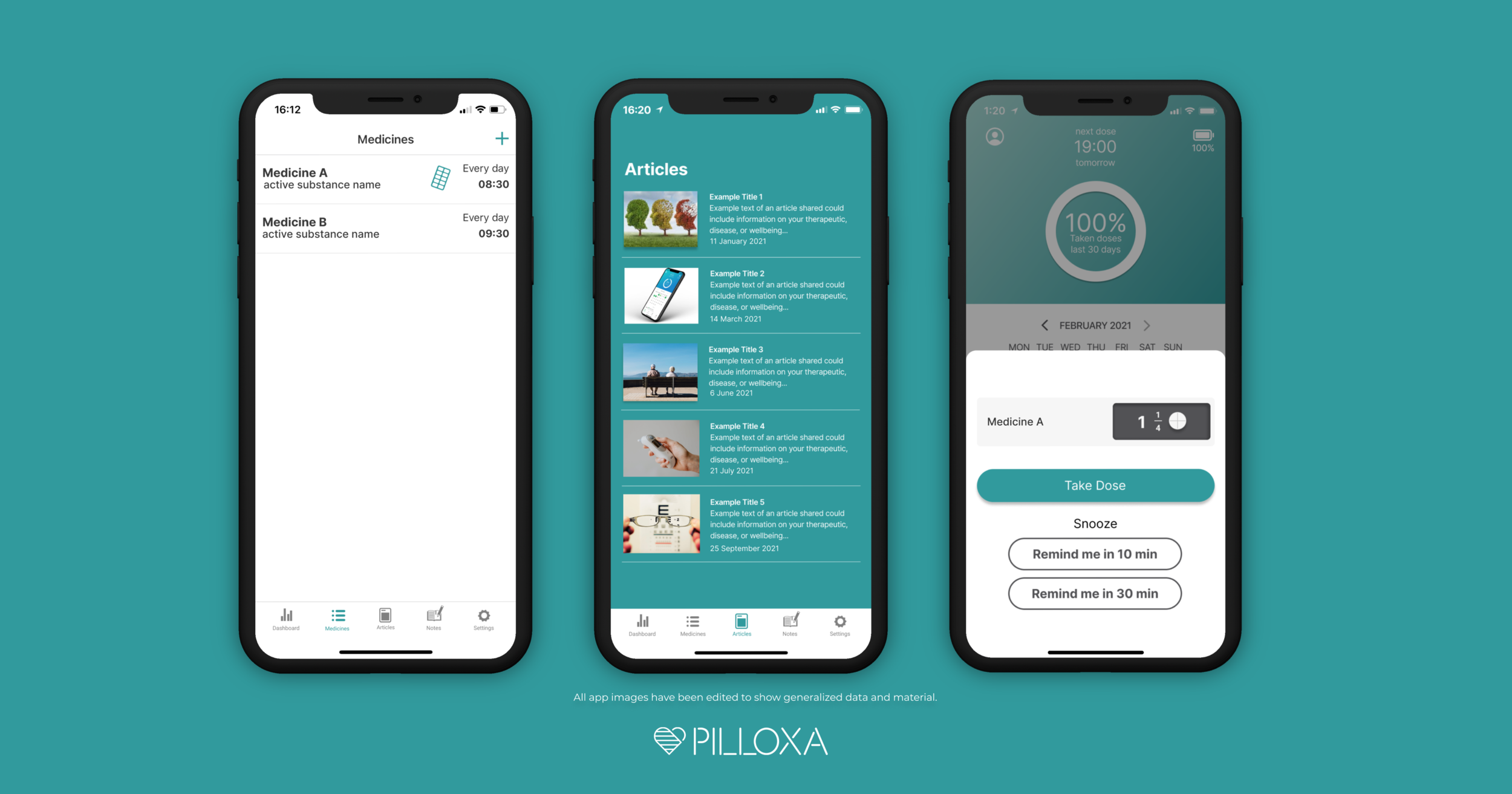
Pilloxa launches the first plug-and-play platform to create apps to connect patients with pharma and HCPs
Stockholm, Sweden, June 3rd, 2021: Digital health company Pilloxa today announced the launch of their new product offering, a cloud-hosted solution for pharmaceutical companies and medical research teams to engage patients and learn from them. Through creating branded programs within Pilloxa’s patient-facing mobile app, the customer can provide patients with a customized experience for a…
Digital health company Pilloxa and University of Oslo start collaborative trial to further validate their smart medication adherence system in a real-world clinical setting
Stockholm, Sweden: Digital health company Pilloxa today announced the start of ASTORIA* – a single-armed and multicenter observational clinical trial in which their digital patient-centric adherence solution will be tested in patients with cardiovascular disease, namely newly diagnosed patients with atrial fibrillation. The study, to be conducted as a multicenter trial with the University of Oslo…
7 causes of medication nonadherence
All of us have forgotten to take a pill. We are human and make mistakes. Some of us neglect to take our medication for accidental reasons, while for others it may be intentional. So, what are the main causes of medication non-adherence? By better understanding the causes, we can work to provide solutions for our patients…
Does medicine really help?
The importance of medication adherence in healthcare and how it impacts society. Written by Helena Rönnqvist, Chief Science Officer at Pilloxa. The “western” world is a knowledge-based world. We have let go of religion, instead championing data and science as the correct and single source of truth. We believe in the individual and health as…
Digital health company Pilloxa receives €580k from EU to develop patient medication adherence system
Company aims to validate a digital solution within the cardiovascular field and to further develop it together with leading Nordic university hospital Stockholm, Sweden: Digital health company Pilloxa today announced that they have received €580k in funding from the EU’s EUROSTARS program. The company is developing digital patient-centric solutions and will, together with the University of…
A clinical trial from the patient side during COVID-19
At Pilloxa, we are bringing digital and patient-centric solutions to clinical studies. My job makes me very curious about the healthcare industry, and it was therefore thrilling for me to experience a clinical trial firsthand last month in the midst of a pandemic unlike anything we have seen during our lifetimes. I want to take…
Continue Reading A clinical trial from the patient side during COVID-19
Bonnier Ventures leads a USD 1.3 million investment in Pilloxa for European expansion
Pilloxa is delighted to announce that Bonnier Ventures, the venture arm of the Bonnier group, has taken the lead in the USD 1.3 million investment round in the company. Pilloxa, founded in 2015, tackles poor medical adherence within the clinical studies industry by having developed a patient centric adherence platform consisting of smart hardware, tailored…
Sandoz and Pilloxa in collaboration using smart pillbox to improve transplant patient medication adherence
Swedish digital startup company Pilloxa today announced that it is to collaborate with Sandoz Nordics on using Pilloxa’s smart pillbox in the field of transplantation. The initial phase aims to support Swedish transplant clinics with Pilloxa’s innovation in order to facilitate improved medication adherence by transplanted patients. Pilloxa originates from the Karolinska Institutet, Stockholm County…
Pilloxa initiates a strategic partnership with Bayer AB for innovative smart pillbox to tackle growing problem of patient compliance
Swedish digital health start-up Pilloxa has entered a strategic collaboration with Bayer AB. Pilloxa helps patients take their medicine in time by using an app that is connected to a smart pillbox. Non-compliance, with patients either forgetting or failing to complete complete courses of medication leads to thousands of deaths and billions of Euros in…
Pilloxa Singled Out as a Great Innovation from Stockholm
The Stockholm Region has presented Pilloxa as one of the Great Innovations within Healthcare. An article has been published in the Swedish magazine “Goda Innovationer”. Read more here: http://sllinnovation.se/artikel/nytt-magasin-om-smarta-innovationer
Continue Reading Pilloxa Singled Out as a Great Innovation from Stockholm
Pilloxa Raises 5,6 MSEK from Prominent Angel Investors
Pilloxa has successfully raised 5,6 MSEK from swedish angel investors, ranging from the previous Global Marketing Director at Spotify Sophia Bendz, the previous CEO of Ratos Susanna Campbell, the founder of Serendipity Innovations Saeid Esmaeilzadeh and the business angel Eva Redhe. The funding will be utilized to clinically validate the product together with some of…
Continue Reading Pilloxa Raises 5,6 MSEK from Prominent Angel Investors
Pilloxa Receives Funding from the European EIT Health!
Pilloxa is one of the 10 companies in Scandinavia that was awarded the EIT Health Headstart funding of € 50 000. EIT Health is the life science division of EIT which is an EU organization that spurs innovation and entrepreneurship across Europe to overcome some of its greatest challenges. The funds will be used to…
Continue Reading Pilloxa Receives Funding from the European EIT Health!
Pilloxa Receives Funding from Vinnova!
Pilloxa is one of the companies awarded a grant from the Swedish innovation agency Vinnova. (article in swedish) Out of 138 applicants, Pilloxa is one of the companies that was awarded funding from the Swedish innovation agency Vinnova. The funds from “Innovationsprojekt i företag” will be used to verify the business model of Pilloxa in…
Pilloxa Mentioned on the Stockholm County Council Innovation as One of the Most Inspirational Innovation Cases
The healthcare organization in Stockholm, Stockholm County Council (SLL) has an innovation division where they list inspirational cases of innovations that have spun out of SLL. Pilloxa is listed as the first project on this list and have also received funding two times from the innovation fund within SLL. For more info, see the following…
Pilloxa Receives CE Marking
Pilloxa has received a certificate of registration from the Swedish Medical Products Agency (Läkemedelsverket). Pilloxa is now marked as a medical device class I and can therefore now be marketed in the EU.